Authors / metadata
DOI: 10.36205/trocar7.2026002
Abstract
Background: Stress urinary incontinence (SUI) and mixed urinary incontinence (MUI) are prevalent conditions among women, significantly impacting quality of life. While midurethral sling procedures remain the gold standard, concerns over mesh-related complications have driven interest in minimally invasive alternatives such as urethral bulking agents. Bulkamid®, a polyacrylamide hydrogel, is increasingly used as a first-line or adjunctive treatment.
Objectives: To evaluate the current evidence regarding the efficacy, safety, durability, reinjection rates, and comparative outcomes of Bulkamid® in the treatment of female SUI and MUI.
Evidence Acquisition: A comprehensive narrative review was conducted using data from randomized controlled trials, prospective cohort studies, systematic reviews, and long-term follow-up studies involving women treated with Bulkamid® for SUI or MUI. Sources were identified through PubMed and Cochrane Library searches.
Results: Subjective symptom improvement rates following Bulkamid® injection ranged from 60% to 80%, with objective cure rates of 40% to 60% at 12 months. Long-term durability has been demonstrated in follow-up periods extending up to 7 years. Reinjection was required in approximately 25%–30% of cases. The safety profile is favorable, with a low incidence of serious adverse events. Most complications are minor and transient, such as urinary retention or urinary tract infection. Compared with other urethral bulking agents and midurethral slings, Bulkamid® shows lower efficacy than slings but superior safety and tolerability.
Conclusions: Bulkamid® represents a viable, minimally invasive treatment option for select women with SUI or MUI, particularly those seeking to avoid surgical intervention. Its favorable safety profile and durability make it a reasonable alternative in appropriately selected patients. Further randomized head-to-head trials and cost-effectiveness analyses are warranted.
Introduction
Female urinary incontinence, particularly stress urinary incontinence (SUI) and mixed urinary incontinence (MUI), represents a significant global health challenge, affecting up to 50% of women and leading to considerable psychosocial and economic burdens (1) SUI is typically caused by urethral hypermobility or intrinsic sphincter deficiency (ISD), while MUI involves overlapping symptoms of urgency and stress leakage, complicating both diagnosis and therapeutic approaches (2). While mid-urethral sling (MUS) procedures have been widely considered the gold standard for SUI management, increasing scrutiny over mesh-related complications – including erosion, chronic pelvic pain, and regulatory concerns – has fueled a resurgence of interest in urethral bulking agents (UBAs) as a safer, less invasive alternative (3). Among the available UBAs, Bulkamid®, a polyacrylamide hydrogel approved by the FDA in 2020, has emerged as a promising second-line treatment owing to its biocompatibility, ease of administration, and sustained symptom relief (4). This literature review critically synthesizes current evidence on the efficacy, safety, durability, and comparative performance of Bulkamid®, situating its role within the evolving landscape of SUI and MUI management.
Results
Efficacy and Clinical Outcomes of Bulkamid®
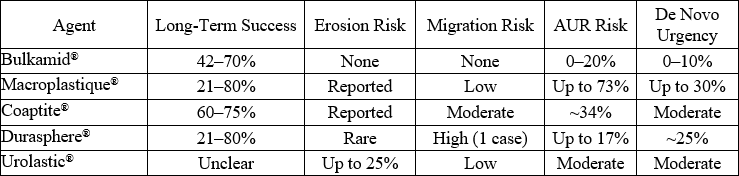
Bulkamid® exerts its therapeutic effect by enhancing urethral coaptation through submucosal injections, thereby supporting the urethral sphincter during increases in intra-abdominal pressure. The majority of prospective studies report symptom improvement rates between 60% and 80%, with objective cure rates ranging from 40% to 60% at 12 months post-procedure (5). In a pivotal multicenter trial by Mohr et al., 135 women treated with Bulkamid® demonstrated a 66% improvement in symptoms and a 44% objective cure rate after one year (6). A meta-analysis by Hoe et al. (2021), encompassing 1,583 women across 17 studies, found short-term success rates between 29.8% and 89.7%, and long-term efficacy from 42% to 70%, establishing Bulkamid® as a reliable and consistent option across diverse populations (7). Critically, while Bulkamid® does not outperform MUS in terms of absolute cure rates (66.4% vs. 95% in some randomized trials), it presents a compelling balance between moderate efficacy and superior safety, particularly for women seeking non-surgical solutions (8).
Objective Outcome Measures and Patient Satisfaction
Multiple studies highlight improvements in quantifiable incontinence metrics, including: Pad weight reduction and significant improvements in International Consultation on Incontinence Questionnaire (ICIQ) scores (5). High levels of patient satisfaction, with VAS scores exceeding 80/100 in most trials, emphasizing the favorable subjective experience even when objective cure is incomplete (6-9). These findings suggest that Bulkamid® not only addresses the clinical burden of incontinence but also aligns with patients’ quality-of-life expectations, an increasingly prioritized outcome in modern urogynecological care.
Safety Profile and Complication Rates
Unlike earlier-generation UBAs, which often comprised particulate materials associated with migration, granuloma formation, and erosion, Bulkamid® is a non-particulate, hydrophilic hydrogel that remains localized at the injection site. It demonstrates minimal immunogenicity, with rare reports of serious adverse events (4). Urinary tract infection (UTI) rates ranged from 1.6% to 40%, with acute urinary retention (AUR) reported in up to 20% of patients—though most cases were transient and resolved without intervention (10). Compared to tension-free vaginal tape (TVT), Bulkamid® had significantly lower reoperation (0% vs. 6%) and serious complication rates, including erosion, hematoma, and chronic retention (11). These data reinforce Bulkamid®‘s position as the safest available UBA, particularly for populations at elevated risk for surgical or mesh-related complications (12) .
Durability and Reinjection Rates
Durability of response is a key consideration in minimally invasive interventions. Although some patients require reinjection, studies consistently report that bulk volume remains stable over time, and the need for repeat procedures should be interpreted within the framework of individualized care rather than as treatment failure. Toozs-Hobson et al. (2021): Over a 7-year follow-up, 67.1% of women reported sustained symptom relief; 24.2% required one reinjection, and 6.5% needed two (12). Agerskov et al. (2011) and Lose et al. (2010) found reinjection rates between 22.5% and 26%, typically within the first 3–6 months (14,15). FDA SSED (2020) data reported a reinjection rate of ~30%, affirming real-world observations (16). Despite this variability, durable symptom control is achievable in the majority, especially when reinjection is part of an adaptive treatment strategy (17,18).
Discussion
Overall, the literature supports Bulkamid® as a safe, minimally invasive, and moderately effective treatment option for women with SUI. Its favourable safety profile, stability, and patient satisfaction make it particularly useful for select populations, including those unfit for surgery or seeking alternatives to mesh-based procedures. The use of urethral bulking agents has re-emerged as a viable therapeutic option for women with stress urinary incontinence (SUI), particularly in those seeking minimally invasive approaches or who are poor candidates for surgery. Among available agents, Bulkamid®, a non-particulate polyacrylamide hydrogel (PAHG), has gained prominence due to its favourable safety profile, durability, and ease of administration. The literature consistently demonstrates that Bulkamid® provides meaningful symptom improvement, though often with lower objective cure rates than midurethral sling procedures. In terms of efficacy, Bulkamid® offers moderate to high rates of patient-reported improvement, though “cure” rates vary widely depending on study design, follow-up duration, and evaluation criteria. Patient selection remains a key determinant of success. Bulkamid® appears particularly effective in women with mild to moderate SUI, isolated urethral hypermobility or intrinsic sphincter deficiency (ISD), though reported outcomes in ISD populations remain mixed. Its role as a first-line therapy versus a secondary option after failed sling procedures continues to be explored. Recent studies suggest that Bulkamid® may serve as a salvage treatment for recurrent SUI following mesh complications or sling removal, with acceptable rates of symptom improvement. Another theme in the literature is the quality-of-life benefit associated with Bulkamid®. Even in cases where complete continence is not achieved, patients often report meaningful improvements in daily function and satisfaction. This highlights the importance of patient-centred outcome measures in evaluating bulking agents and supports the view that Bulkamid® can be a valuable component of the therapeutic arsenal, particularly when treatment goals emphasize improvement rather than cure.
Conclusion
Bulkamid® represents a clinically validated, patient-centered and minimally invasive option for women with SUI or MUI, offering durable efficacy, high safety and excellent tolerability. Its superiority over other UBAs, in terms of biocompatibility and adverse event profile, positions it as a first-line non-surgical therapy for selected patients. While not a replacement for mid-urethral slings in all cases, its role is increasingly recognized in personalized, guideline-driven urologic care. With further comparative trials and evidence- based guideline updates, Bulkamid® is poised to reshape the landscape of female incontinence treatment.
Future Directions
To optimize the clinical positioning of Bulkamid®, the literature emphasizes several research imperatives:
- Standardization of outcome definitions to facilitate comparative studies.
- Head-to-head RCTs among UBAs to establish definitive efficacy hierarchies.
- Development of predictive biomarkers for response and reinjection need.
- Longitudinal studies on cost-effectiveness, particularly in elderly populations.