Authors / metadata
DOI: 10.36205/trocar7.2026001
Abstract
Women presenting at an older reproductive age or with premature ovarian insufficiency pose significant challenges for fertility specialists. Among the available options – tailored stimulation protocols for poor responders, adjuvants such as growth hormone or micronutrients, and lifestyle measures – there is a growing need for more result-oriented treatments. Ovarian rejuvenation with platelet-rich plasma (PRP) may offer improved prospects in both age-related decline and premature ovarian failure. PRP is derived from autologous blood after removal of red and white cells and contains platelets enriched with cytokines and growth factors. In addition to its hemostatic function, PRP delivers the growth factor proteins: VEGF, PDGF, TGF-β and IGF-1, which are synergically implicated in angiogenesis, stromal remodeling and follicular activation (1). PRP has long been applied in dermatology, orthopedics and regenerative medicine. Its intraovarian use is based on the presence of stem-like cells within ovarian tissue. Recent data suggest that these stem cells may contribute to neo-oogenesis or enhance responsiveness to gonadotropin stimulation when exposed to growth-factor-rich environments (2).
Introduction
PRP can be injected in the ovaries with an Ovum Pick Up (OPU) needle by Transvaginal ultrasound guidance or laparoscopically with a laparoscopic injection needle. Both are wide bore needles and chances of spillage is much more owing to the small sized ovaries in patients with poor ovarian reserve. The good results of suprapubic approach are highlighted by using a spinal needle suprapubicaly with the laparoscope in place and injecting at 2-3 places in the ovary under vision making sure there is no spillage.
Material and Methods
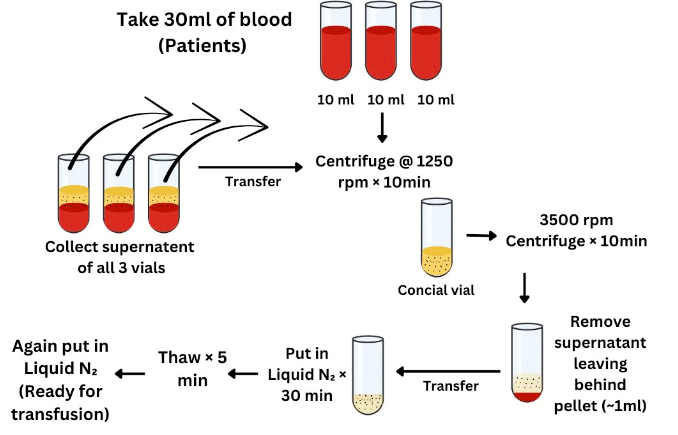
Thirty milliliters of venous blood were collected in three 10 ml conical tubes. Samples were centrifuged at 150 × g for 10 min. The supernatants were pooled and centrifuged again at 3500 × g for 10 min. The concentrate was transferred into a cryovial, frozen in liquid nitrogen for 30 min, thawed for 5 min and re-frozen (figure 1). The preparation was gently warmed prior to injection. Double-spin protocols are widely considered superior for achieving higher platelet concentrations, which may enhance growth-factor delivery. Timing between activation and intraovarian injection appears critical, as growth-factor release is highest within hours of platelet activation (3).
Surgical Technique
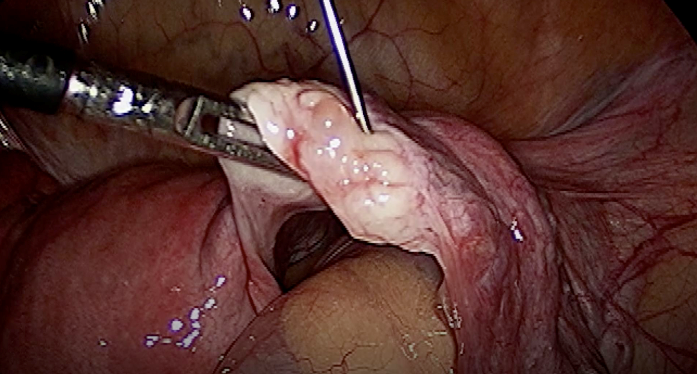
A five mm laparoscope and a four mm secondary trocar were introduced. The ovary was mobilized to the suprapubic midline. Using an 18-gauge spinal needle, approximately two ml PRP was injected into each ovary at 1–2 cortical sites. Patients were discharged within 4–6 h. The use of a spinal needle offers three advantages: 1) reduced cortical trauma compared to OPU needles, 2) minimized PRP spillage into the peritoneum and 3) improved targeting of the ovarian cortex, where primordial follicles are located. This approach is particularly valuable in small, atrophic or fibrotic ovaries (figure 2) (4).
Results
This non-randomized interventional study involved 23 women with poor ovarian response and previous failed IVF attempts. 17 women underwent laparoscopic intraovarian PRP injection; six opted for oocyte donation. Those treated showed improved antral follicle count (AFC), oocyte and embryo quality, 11 positive β-hCG tests and seven ongoing pregnancies. Similar observational studies reported increases in AMH, reductions in basal FSH and improved embryo development after intraovarian PRP (5).
Discussion
The results of this work support intraovarian PRP as a feasible intervention with potential benefit for poor responders. The distinctive element of our approach is the use of the 18G spinal needle. This modification improves cortical precision, reduces leakage and minimizes trauma, thereby enhancing consistency and potentially clinical outcomes. Growth factors in PRP promote endothelial proliferation, improve stromal vascularity and create a more favorable environment for follicular growth. These paracrine effects may also improve granulosa cell function, mitochondrial activity and recruitment of dormant follicles. Similar observational studies reported increases in Anti Müllerian Hormone (AMH), reductions in basal FSH and improved embryo development after intraovarian PRP (6).
Conclusion
Intraovarian PRP injected with a thinner 24/25-gauge spinal needle minimizes extravasation and leakage of PRP and has given good oocyte retrieval numbers and live birth rate. It has to be taken into account that this report represents a very small retrospective study.
Editor’s note
We are publishing this article because, in our view—and as confirmed by the reviewers—it contains noteworthy content. It should be noted that this is an intervention study that does not meet standard scientific criteria. Inclusion criteria and an ethics committee approval are not provided. However, the findings published here should be made available as a basis for further clinical studies.